Evidence Requiring an Explanation
Experimental Support. Good theories have considerable experimental support.
1.
HP: Every phenomenon involved in the hydroplate explanation for Earth’s radioactivity is well understood and demonstrable: the piezoelectric effect, poling, nuclear combustion, heat absorption by fusion of heavy elements, electron capture, flutter with high compressive and tensile stresses, neutron production by bremsstrahlung radiation, Z-pinch, neutron activation analysis, rapid decay of artificially produced superheavy nuclei, and increased decay rates resulting from high voltages and concentrated electrical currents.
We know radioactive nuclei have excess energy, continually vibrate, and are always on the verge of “flying apart” (i.e., decaying). Atomic accelerators bombard nuclei; adding that energy produces radioisotopes and rapid decay.
2.
CE: We are dealing with variables such as temperature time, and size) in and around stars that are hundreds of thousands of times more massive than here on Earth. Therefore, experimental support for chemical evolution is necessarily limited. Experiments using particle colliders allow investigation of the interactions of subatomic particles traveling at great speeds. By using computer simulations and extrapolating the results of experiments to larger scales, we can infer of elements that would have been produced at extremely high temperatures inside huge stars billions of years ago.
Quartz Alignment in Continental Crust. Why are quartz crystals aligned in most quartz-rich rocks? 85
3.
HP: As explained in Figure 213 on page 393, electric fields, from centuries of cyclic compression and tension (twice a day) before the flood, increasingly aligned quartz crystals in granite—a process called poling. Amazingly, laboratory tests have shown that alignments still exist even after the compression event and thousands of years.85
4.
CE: Electrical fields must have been present as Earth’s rocks solidified from a melt. The electrical fields would have aligned the quartz grains.
[Response: Granite consists of a mixture of millimeter-size mineral grains. The isolated quartz crystals we see today, would not have formed if the granite crust slowly cooled and solidified from a melt—even if a strong electrical field had been present. As the melt slowly cooled, each type of mineral would eventually solidify and either sink or float (depending on its density), thereby sorting into thick layers and very large crystals, such as pegmatites. Rapid cooling would have produced a rock called rhyolite. Granite cannot form from a melt.]
Radioactivity Concentrated in Continental Crust. Why is Earth’s radioactivity concentrated in the continental crust?
5.
HP: During the flood, powerful electrical discharges within the fluttering granite crust produced Earth’s radioactivity. Therefore, Earth’s radioactivity should be concentrated in the continental crust.
Earth’s ocean floors and mantle have little radioactivity, because they did not flutter and they contain little to no quartz, so they could not produce strong electrical discharges. Also, the subterranean water absorbed most neutrons generated in the fluttering crust, so there is little radioactivity below Earth’s crust.
6.
CE: Stars produced radioisotopes. Later, Earth formed from the debris of exploded stars—“starstuff.” What concentrated Earth’s radioactivity in the continental crust is unclear.45
[Response: If Earth formed from the debris of exploded stars, radioactivity should be distributed throughout the Earth, not concentrated in the crust.]
Correlation of Heat Flow with Radioactivity. Heat flowing out of the Earth at specific continental locations correlates with the radioactivity in surface rocks at those locations.
7.
HP: Electrical discharges within the crust generated both heat and radioactivity. The more electrical current at a location, the more radioactivity and heat produced. Therefore, the heat flowing up through the Earth’s surface should correlate with radioactivity at the Earth’s surface.
8.
CE: The following may explain this correlation:
- slow radioactive decay generated some of the heat flowing out of the Earth,
- each vertical column immediately below Earth’s surface has a different but uniform amount of radioactivity,
- radioactivity varies widely over horizontal distances as short as 50 miles, and
- enough time has passed to conduct most of that deep heat up to the surface.
If so, radioactivity goes only 4.68 miles down.119 If it went much deeper, the heat coming out at the surface, after just a few million years of radioactive decay, would be much more than is coming out today.
Although it is unlikely that all radioactivity is concentrated in Earth’s top 4.68 miles, radioactivity may decrease with depth, allowing even more time (consistent with the great age of the Earth) for that deeper heat to flow to the surface. Millions of such variations could be imagined, but all visualize radioactivity as being concentrated near the surface.
[Response: Millions of years would be required for the heat to flow up 4.68 or more miles.120 If that much time elapsed, some locations would have eroded more than others. Arthur Lachenbruch has shown that millions of years of surface erosion would destroy the correlation unless radioactivity decreased exponentially with depth.121 If so, too much time would be required for the deeper heat generated to reach the surface. However, Germany’s Deep Drilling Program found that variations in radioactivity depended on the rock type, not depth.122]
Ocean-Floor Heat. Continental (granitic) rocks have much more radioactivity than the ocean floors, so why is slightly more heat coming up through the ocean floors than through the granite continents?
9.
HP: During and soon after the flood, deep frictional deformation below the ocean floors produced heat that is coming up through the ocean floors today. [See “Magma Production and Movement” on page 156.] The granite crust contains almost all of Earth’s radioactivity, because piezoelectric effects in the fluttering crust released powerful electrical discharges within granite and generated Earth’s radioactivity.
10.
CE: Much of the heat coming up from within the Earth is produced by radioactive decay. Yet, Stacey has admitted:
The equality of the continental and oceanic heat flows is puzzling in view of the great disparity in the total amounts of the radioactive elements uranium, thorium, and potassium in the continental [granitic] and oceanic [basaltic] crusts.123
[Response: Stacey’s data actually show that the oceanic heat flow is slightly greater than that coming up through the continents.]
Argon-40 ( 40Ar). Today, 40Ar is produced almost entirely by the decay of potassium-40 ( 40K) by electron capture. Earth does not appear to have enough 40K to produce all the 40Ar in our atmosphere—even if the Earth were twice as old as evolutionists claim. Saturn’s moon, Enceladus, also has too much 40Ar but not enough 40K.
11.
HP: 40K was produced in several ways as the crust was fluttering during the global flood. Z-pinching from the powerful electrical surges produced superheavy elements. Because they were all too proton-heavy, they quickly fissioned into thousands of isotopes, including radioactive isotopes. Some would have been 40K.
40K was also produced in other ways. Calcium is the fifth most abundant element in the Earth’s crust, 97% of which is calcium-40 (40Ca). Most calcium came from the subterranean chamber, the source of Earth’s vast limestone (CaCO3) deposits. [See “The Origin of Limestone” on pages 258–264.] Each 40Ca nucleus that captured an electron during the electrical surges became 40K.
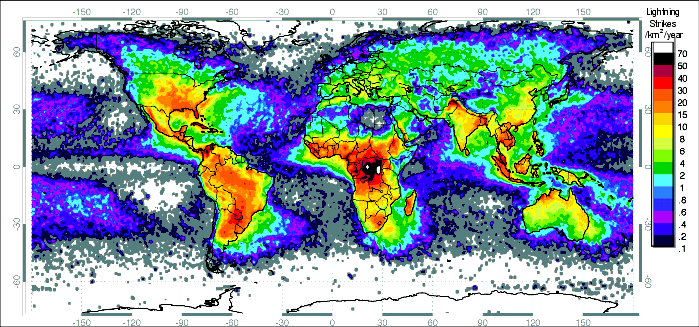
Figure 218: Lightning Frequency. Today, more lightning strikes occur along the equator in central Africa than anywhere else on Earth— more than 100 strikes per square kilometer each year. The center of this region is only about 1000 miles east of Oklo. Probably more violent electrical storms occurred farther to the west soon after the flood, as warmer moist air rising off the Atlantic collided with the cold air above the temporarily high continent of Africa.
Regardless of how 40K formed, it would have become 40Ar by capturing an electron during the electrical surges in Earth’s fluttering crust. Consequently, 40Ar was produced almost simultaneously with the production of 40K. (Argon is a nobel gas, so none of its 24 isotopes react chemically with other elements.) Much of the abundant 40Ar was able to escape into the atmosphere, so today 40Ar is the third most abundant gas in Earth’s atmosphere (not counting water vapor).
Today, about 5,000 years after the flood and that electrical storm in Earth’s crust, 40K rarely captures an electron, so 40K decays slowly to 40Ar with a half-life of 1.3-billion years. Those who do not understand how almost all 40K and 40Ar were rapidly produced during the flood, frequently find much 40Ar alongside 40K. They argue that it was produced by the slow decay of 40K after molten rock solidified, because argon would have bubbled out of any molten material. But molten rock produced during the flood (and therefore under water and pressure) would not have been able to release its dissolved 40Ar. Molten rock in contact with liquid water would instantly form a crust at the water-rock interface that would prevent 40Ar’s escape.
So one might mistakenly think that rock was billions of years old if they dated it by the potassium-argon dating technique, because it contained so much argon that today only builds up slowly when 40K decays with a half-life: of 1.3-billion years. Actually the rock formed soon after the flood began in 3290 B.C [See page 485.]
As for lava flows that have occurred since the flood, the potassium-argon dating technique is seldom used if the rock is thought to be younger than 100,000 years.
12.
CE: The argon on Enceladus needs to be remeasured.
Crustal rocks contain little potassium-40, but the mantle may contain much more. Furthermore, if about 66% of the mantle’s 40Ar escaped into the atmosphere, both the atmosphere’s 40Ar and the needed 40K in the Earth’s crust and mantle could be explained.124
[Response: This 66% proposal is ridiculous, because argon, a large atom, is easily trapped between mineral grains and within crystal structures. Indeed, the potassium-argon dating method is used, because solids retain argon over long periods.]
Oklo Natural “Reactor.” Can Oklo be explained? Why haven’t other uranium deposits become nuclear reactors?
13.
HP: Today, a region near Oklo receives more lightning strikes than anywhere else on Earth. [See Figure 218] For centuries after the flood, warm oceans and heavy precipitation (explained on page 134) would have generated even more frequent and severe thunderstorms. As lightning strikes passed down through the thin layer of uranium ore, bremsstrahlung radiation125 accelerated radioactive decay and released neutrons, as explained on pages 390 and 398. Those neutrons then fissioned 235U and initiated brief, subcritical chain reactions. Their consequences are detected in isolated zones within 30 kilometers of the Oklo mine.
Lightning strikes would also explain why the ratio of 235U to 238U at Oklo varied a thousandfold over distances of less than a thousandth of an inch.55 Lightning branches successively into thousands of thin, fractal-like paths, some quite close together.
14.
CE: Today, 0.72% of natural uranium is 235U. Because 235U decays faster than the more abundant 238U, a higher percentage of uranium would have been 235U in the past. About 2-billion years ago, 3.7% of all uranium worldwide would have been 235U, enough for uranium deposits to “go critical” if other factors were favorable. One important factor is having water saturate the uranium ore. If the ore “went critical” and heated up, the water would evaporate, so the reactor would shut down and cool off. This cycle may have repeated itself many times. When the Earth’s crust solidified at least 3.8-billion years ago, even more 235U was concentrated. Why hundreds of other uranium ore deposits did not become natural reactors is a mystery.
[Response: Such cycles would not produce temperature variations and power surges as extreme as Harms found them to have been.56 Certainly, we would not expect to see thousandfold variations in the ratio of 235U to 238U over distances of less than a thousandth of an inch, especially after 2-billion years.
Disposal of radioactive waste from nuclear reactors is a serious environmental problem. Few believe that any geological formation can contain radioactive waste for 100,000 years—even if held in thick, steel containers encased in concrete. However, at Oklo, most products of 235U decay have not migrated far from the uranium deposit,127 despite 2-billion years of assumed time.]
Helium-3 ( 3He). 3He production begins with a nuclear reaction that yields 3H, which then beta decays to 3He.41 So why is 3He common inside the Earth, why are black smokers expelling large amounts of 3He, and why does the ratio of 3He to 4He (neither of which decays) vary so widely inside the Earth?
15.
HP: During the flood, many nuclear reactions occurred inside the fluttering crust and in the porous floor of the subterranean chamber. Today, black smokers expel 3He and SCW from that porous floor. 3He also escapes to the Earth’s surface along faults in the crust, so the amount of 3He varies widely at different locations.
16.
CE: Nuclear reactions seldom occur inside the Earth, so 3He must be primordial—originating from the very beginning (the big bang).128 The Earth grew and evolved by meteoritic bombardment. Therefore, 3He was brought to the Earth as it evolved by meteoritic bombardment.
[Response: The premise that “nuclear reactions seldom occur inside the Earth” is wrong. The hydroplate theory explains how nuclear reactions occurred inside the fluttering crust during the flood, thus producing 3He.
How could helium, a light, inert gas, have been trapped in meteoritic material or in a supposedly molten Earth, where it would bubble to the surface?42 Even if helium became trapped in an evolving Earth, why would the ratio of 3He to 4He vary so widely from location to location? If the mantle is circulating, as textbooks erroneously teach, the small amount of 3He should be so diluted it would be undetectable.44
One theory, which has gained little support, claims that a natural uranium reactor, 5 miles in diameter, has been operating at the center of the Earth for 4.5-billion years. The lighter fission products from that reactor, such as 3He, supposedly migrated up 4,000 miles, primarily through solid rock. One problem with this idea is that any 3He produced near a neutron source would readily absorb a neutron and become 4He. The hypothetical reactor would provide those neutrons, as would any fissioning material (such as uranium or thorium) near the 3He’s 4,000-mile upward path. Likewise, 3He atoms that somehow fell to the Earth 4,500,000,000 years ago would have to avoid free neutrons for a long time.]
Zircon Characteristics. Why do zircons found in Western Australia contain strange isotopes and microdiamonds?
17.
HP: Inside these zircons, more uranium and thorium decayed than almost anywhere else on Earth. If that decay always occurred at today’s rates, as evolutionists maintain, then those zircons formed back when the Earth was probably too hot to form zircons—a logical contradiction. Therefore, at some time in the past, decay rates must have been much faster.
The compression event or shock collapse (explained on page 392) likely produced the high pressures required to form microdiamonds. Minerals and isotopes in these zircons show that water and granite were also present.38 The low ratio of 13C to 12C suggests that all these carbon isotopes were not originally present. Therefore, at least some carbon isotopes were produced or consumed, and that implies nuclear reactions. These zircons and their contents probably formed in the plasma channels “drilled” by the electrical discharges at the beginning of the flood.
18.
CE: Organic matter contains low ratios of 13C to 12C. Therefore, the presence of water and the low ratio of 13C to 12C could imply that life was present on Earth long before we evolutionists thought.
Although the Earth was extremely hot 4.0–4.4-billion years ago, some regions, such as above ocean trenches, where the geothermal heat flow is up to 17% lower than normal, must have been cold enough to crystallize zircons.129 If so, plate tectonics operated two billion years before we thought, although ancient trenches have never been found. [See “ ‘Fossil’ (Ancient) Trenches” on page 176.]
Helium Retention in Zircons. Based on today’s slow decay rates of uranium and thorium in zircons, some zircons are claimed to be 1.5-billion years old, but their age, based on the diffusion of helium out of those same porous zircons, was only 4,000–8,000 years.40
19.
HP: About 5,000 years ago, electrical discharges within the crust produced accelerated decay (1) during the weeks the crust fluttered at the beginning of the flood, and (2) during the sudden compression event near the end of the flood. Helium produced by the decay of uranium and thorium in zircons, is still diffusing out. Very little helium has escaped from zircons, because little time has passed since the flood. [See "Helium" on page 38.]
20.
CE: Only a few helium diffusion rates in zircons have been measured. Besides, those few measurements were not made under the high pressures that exist 1–2 miles inside the Earth. Helium cannot escape rapidly through cracks in zircons under high pressures, so closed cracks could explain why so much has been retained in 1.5-billion-year-old zircons. If the diffusion rates measured in the laboratory are 100,000 times too high, the discrepancy would be explained.
[Response: Such large errors are unlikely, and hard, tiny zircons have few cracks, even at atmospheric pressure.]
Isolated Polonium Halos. Polonium-218, -214, and -210, ( 218Po, 214Po, and 210Po) decay with half-lives of 3.1 minutes, 0.000164 seconds, and 138 days, respectively. Why are their halos found without the parents of polonium?
21.
HP: During the early weeks of the flood, electrical discharges throughout the fluttering crust produced thin plasma channels in which superheavy (extremely unstable) elements formed. Then, they quickly fissioned and decayed into many relatively lighter elements, such as uranium.
Near the end of the flood, the compression event crushed and fractured rock, producing additional piezoelectric discharges, bremsstrahlung radiation and accelerated radioactive decay. Hot SCW (held in the spongelike voids in the lower crust) and 222Rn (an inert gas produced in plasma channels) were forced up through these channels and fractures. As the mineral-rich water rose hours and days later, its pressure and temperature dropped, so minerals, such as biotite and fluorite, began forming in the channels. Wormlike myrmekite also formed as quartz and feldspars precipitated in the thin, threadlike channels “drilled” by the powerful electrical discharges and by SCW (a penetrating solvent).
In biotite, for example, why were a billion or so polonium atoms concentrated at each point that quickly became the center of an isolated polonium halo? Why didn’t each halo melt in minutes as hundreds of millions of alpha particles were emitted? In a word, water.
Biotite requires water to form. Within biotite, water (H2O or HOH) breaks into H + and OH -. The OH - (called hydroxide) occupies trillions upon trillions of repetitive positions within biotite’s crystalline structure. Other water (liquid and gas) transported 222Rn (which decayed with a half-life of 3.8 days) between the thin biotite sheets as they were forming.
Radon gas is inert, so it has no electrical charge. When 222Rn ejects an alpha particle, 5.49 MeV of kinetic energy is released, and 222Rn instantly becomes 218Po with a -2 electrical charge.
Because both energy and linear momentum are conserved, 2% of that energy was transferred to the recoiling polonium nucleus, sometimes embedding it in an adjacent biotite sheet. That recoil energy was so great and so concentrated that it released thousands of hydroxide particles, each with one negative electrical charge.130 Flowing water cooled the biotite and swept away the negatively charged hydroxide. The large number of positive charges remaining quickly attracted and held onto the newly formed polonium flowing by, each with a -2 electrical charge. Minutes later, the captured polonium decayed, expelled more hydroxide, and repeated the process. Within days, these points with large positive charges became the centers of parentless polonium halos. Again, we see that the subterranean water is the key to solving this halo mystery.131 [See "Frequency of the Fluttering Crust" on page 347.]
Similar events happened in other micas and granitic pegmatites. Likewise, the newly formed uranium atoms readily fit in the mineral zircon as it grew, because uranium’s size and electrical charge (+4) fit nicely in the slots zirconium atoms (after which zircons are named) normally fill. Thorium also fits snugly.
Figure 207’s caption (on page 388) states that both the 235U decay series and the 232Th decay series produce other polonium isotopes that decay in less than a second: 215Po and 211Po in the 235U decay series and 216Po and 212Po in the 232Th decay series. However, those isotopes produce few, if any, isolated polonium halos. Why are they missing, when isolated halos from 218Po, 214Po, and 210Po in the 238U decay series are abundant?
Again, radon and water provide the answer. Today, radon (219Rn) in the 235U decay series decays with a half-life of 3.96 seconds, and radon (220Rn) in the 232Th decay series decays with a half-life of 55.6 seconds—82,900 and 5,900 times faster, respectively, than the 3.8-day half-life of 222Rn from the 238U series. Therefore, 219Rn and 220Rn can’t travel far as they look for growing sheets of biotite (or similar minerals) with extremely concentrated, positive electrical charges to attract radon’s negatively charged polonium daughters.
Indeed, as explained on page 388, Henderson and Sparks discovered that the isotopes that produced the isolated halos did flow in channels next to growing biotite sheets, because halo centers tended to cluster in a few sheets but were largely absent from nearby parallel sheets. Therefore, it again appears that certain biotite sheets took on increasing positive charges at specific impact points. Those points then rapidly attracted negatively charged polonium still flowing by. The electrical clustering of polonium, perhaps over days or weeks, produced isolated polonium halos. Later, the high-pressure water escaped, and adjacent sheets were compressed together and weakly “glued” (by hydroxide, a derivative of water) into “books” of biotite.
Collins’ limited deductions, mentioned on page 389, are largely correct, although they raise the six questions italicized below. The hydroplate theory easily answers those questions.
- What was the source of all that hot, flowing water, and how could it flow so rapidly up through rock? Answer: When the flood began, water filled thin, spongelike channels in the lower crust—formed by the tremendous dissolving power of an ocean’s worth of subterranean SCW. Other channels were “drilled” by the powerful electrical discharges and produced by fractures during the compression event. As the high-pressure water rose, the pressure inside the channels increasingly exceeded the confining pressure of the channel walls, so those walls expanded. After the flood, the water cooled and escaped, so the channels slowly collapsed.
- Why was the water 222Rn rich?, because 222Rn has a half-life of only 3.8 days? Answer: As described above, 222Rn’s half-life still allowed it to be widely scattered. Secondly, because it carries no electrical charge, it does not combine chemically with other elements. However, when it encountered liquid water, it went into solution and traveled great distances in the high-pressure flow, usually upward.
- Because halos are in different geologic periods, did all this remarkable activity occur repeatedly, but at intervals of millions of years? If so, how? Answer: The millions of years are a fiction—a consequence of not understanding the origin of Earth’s radioactivity and the accelerated decay processes.
- What concentrated a billion or so 218Po atoms at each microscopic speck that became the center of an isolated polonium halo? The negatively charged polonium atoms were attracted to points with large positive charges. [See “Recoil ” above.]
- Today’s extremely slow decay of 238U (with a half-life of 4.5-billion years) means that today its daughters, granddaughters, etc. form slowly. Were these microscopic specks the favored resting places for 218Po for billions of years, or did the decay rate of 238U somehow spike just before all that hot water flowed? Remember, 218Po decays today with a half-life of only 3.1 minutes. Answer: As the flood began, electrical discharges instantly produced very unstable superheavy isotopes that rapidly fissioned and decayed—similar to the experiments of Dr. Fritz Bosch (in Germany), Dr. Stanislav Adamenko (in Ukraine), and William Barker (in the U.S.A.). The fission and decay products included many new isotopes (such as 222Rn) and heavy chemical elements that did not exist before the flood.
- Why are isolated polonium halos associated with parallel and aligned myrmekite that resemble tiny ant tunnels? Answer: Before the flood, SCW easily dissolved certain minerals in granite (such as quartz and feldspars). During the flood, those hot solutions filled the extremely thin, nearly parallel channels that extended up from the subterranean chamber. After the flood, those solutions rose, evaporated, and cooled, while quartz and feldspars precipitated in some of those channels, becoming myrmekite.
22.
CE: Polonium halos are strange—but only a tiny mystery. Someday, we may understand them.
Elliptical Halos. What accounts for the pair of overlapping 210Po halos in coalified wood in the Rocky Mountains—one halo elliptical and the other spherical, each with the same center?
23.
HP: Some spherical 210Po halos formed in wood that had soaked in water for months during the flood. (Water-saturated wood, when compressed, deforms like a gel.) As the Rocky Mountains buckled up during the compression event, that “gel” was suddenly compressed. Within seconds, partially formed spherical halos became elliptical. Then, the remaining 210Po (whose half-life today is 138 days, about the length of the flood phase) finished its decay by forming a spherical halo superimposed on the elliptical halo.
24.
CE: Only one such set of halos has been found. Again, we consider this only a tiny mystery.
Explosive Expansion. What accounts for the many random fractures surrounding minerals that experienced considerable radiation damage? [See Figure 208 on page 390.]
25.
HP: Radiation damage in a mineral distorts and expands its lattice structure, just as well-organized, tightly-stacked blocks take up more space after someone suddenly shakes them.77 Ramdohr explained how a slow expansion over many years would produce fractures along only grain boundaries and planes of weakness, but a sudden, explosive expansion would produce the fractures he observed.
Accelerated decay, explained on page 398, produced that sudden radiation damage—and heating.
26.
CE: Ramdohr’s observations have not been widely studied or discussed by other researchers.
Uranium-235 ( 235U). If the Earth is 4.5-billion years old and 235U was produced and scattered by some supernova explosion billions of years earlier, 235U’s half-life of 700 million years is relatively short. Why is 235U still around, how did it get here, what concentrated it in ore bodies on Earth, and why do we not see much more lead associated with the uranium? (Observations and computer simulations118 show that few of the 75 heaviest chemical elements—including uranium—are produced and expelled by supernovas!)
27.
HP: During the flood, about 5,000 years ago, electrical discharges (generated by the piezoelectric effect)—followed by fusion, fission, and accelerated decay—produced 235U and most of Earth’s other radioisotopes.
28.
CE: We cannot guess what happened so long ago and so far away in such a hot (supernova) environment.
[Response: Evolution theory is filled with such guesses, but usually they are not identified as guesses. Instead, they are couched in impressive scientific terminology, hidden behind a vast veil of unimaginable time, and placed in textbooks. Radioactive decay can be likened to rocks tumbling down a hill, or air leaking from a balloon. Something must first lift the rocks or inflate the balloon. Experimental support is lacking for the claim that all this happened in a distant stellar explosion billions of years ago and somehow uranium was concentrated in relatively tiny ore bodies on Earth.]
Isotope Ratios. The ratio of the amounts of 234U to 238U in uranium ore deposits is amazingly constant worldwide. One very precise study showed that the ratio is 0.0072842, with a standard deviation of only 0.000017. 132 Other uranium isotope ratios are also constant. How could that be?
29.
HP: Obviously, the more time that elapses between the formation of the various isotopes (such as 234U and 238U) and the farther they are transported to their final resting places, the more varied those ratios should be. The belief that these isotopes formed in a supernova explosion millions of light-years away and billions of years before the Earth formed and then somehow collected in small ore bodies in a fixed ratio is absurd. Besides, powerful explosions would have separated the lighter isotopes from the heavier isotopes.
Some radioisotopes simultaneously produce two or more daughters. When that happens, the daughters have very precise ratios to each other, called branching ratios or branching fractions. Uranium isotopes are an example, because they are daughter products of some even heavier element. Recall that the Proton-21 Laboratory produced superheavy elements that instantly decayed. Also, the global flux of neutrons during the flood provided nuclei with enough neutrons to reach their maximum stability. This explains why uranium isotope ratios in ore bodies are fixed. Had the flux of neutrons originated in outer space, we would not see these constant ratios worldwide. Because these neutrons originated throughout the globe-encircling fluttering granite crust, these fixed ratios are global.
Conclusion: Uranium is the daughter product the of now extinct element-184, the heaviest chemical element detected by man, either directly or indirectly. We will call it Proto-Uranium.
30.
CE: Maybe Uranium didn’t form light years away, billions of years ago.
Carbon-14 ( 14C). Why do radiocarbon dates frequently conflict with dates based on other radiometric dating techniques?
31.
HP: Radiocarbon resides primarily in the atmosphere, oceans, and organic matter. Therefore, electrical discharges within the crust at the beginning of the flood did not affect radiocarbon. However, those discharges and the resulting “storm” neutrons and bremsstrahlung radiation in the crust produced almost all of Earth’s other radioisotopes, disturbed their tenuous stability, and allowed them to rapidly decay—much like a sudden storm with pounding rain and turbulent wind might cause rocks to tumble down a mountainside.
This is why very precise radiocarbon dating—atomic mass spectrometry (AMS), which counts individual atoms—gives ages that are typically 10–1000 times younger than all other radiometric dating techniques (uranium-to-lead, potassium-to-argon, etc.).
32.
CE: That radiocarbon may be contaminated.
[Response: Before radiocarbon’s precision was increased by AMS, some attributed this thousandfold conflict to contamination. Studies have now ruled out virtually every proposed contamination source.25]

Forty Extinct Radioisotopes Forty different radioisotopes each produce unique decay products that are found on Earth, but those radioisotopes are extinct. Based on nuclear experiments, the half-lives of those 40 radioisotopes are so long that they should still be around if the Earth is less than 10,000 years old.133 Does this mean that the Earth is much older than 10,000 years?
33.
HP: That would be true if decay rates had always been what they are today. One must first understand that radioisotopes continually vibrate and all will eventually decay. Electrical surges, produced during the flood’s compression event, generated bremsstrahlung radiation which excited the more unstable radioisotopes, causing them to quickly decay. Therefore, they are not found in nature, but their stable decay products are.
34.
CE: If Earth were less than 10,000 years old, those 40 radioisotopes should still be here, because they would not have had enough half-lives to completely disappear. Only if the Earth were billions of years old, would they all have decayed. This shows that the Earth is billions of years old.
[Response: That explanation shows a lack of understanding of accelerated radioactive decay. See page 398.] Chondrules How did chondrules form?
35.
HP: See “Chondrules” on page 410.
36.
CE: Because chondrules are in meteorites that have even older radiometric ages than Earth, chondrules are the oldest solid material in the solar system. Although chondrules evolved in outer space where temperatures are almost -460°F (492°F below freezing), they required sudden melting temperatures of at least 3,000°F. It is hard to look back that far and determine what could have formed pieces of rock a few millimeters in diameter, quickly melted that rock, and then encased those liquid droplets in other rock.
[Response: The mystery is solved when one understands the origin of Earth’s radioactivity.]
Meteorites. Radioactive decay products in some meteorites require more time to accumulate—at today’s decay rates—than any other rocks ever found in the solar system.
37.
HP: Electrical surges, not time, produced the high concentration of decay products in some meteorites.
During the flood, pillars within the subterranean chamber experienced the most compression and electrical discharges, which, in turn, produced the greatest number of radioactive decay products. Most meteorites originated from crushed pillars, so more decay products formed in meteorites and deep sedimentary and crustal rocks (those that were closer to pillars). This is why radiometric ages generally increase with depth in the crust.
38.
CE: Meteorites have the oldest known radiometric ages in the solar system, so meteorites must have evolved first. This is how we know the Earth evolved from meteorites and the solar system began 4.5-billion years ago.
[Response: How can gas and dust compact themselves into dense black rocks (asteroids and meteoroids) in the weightlessness of space? See “The Origin of Asteroids and Meteoroids” on pages 338–362.]
Close Supernova? Today, half of iron-60 ( 60Fe) will decay into nickel-60 ( 60Ni) in 1,500,000 years. In two meteorites, 60Ni was found in minerals that initially contained 60Fe.142 How could 60Fe have been locked into crystals in those meteorites so quickly,143 that measurable amounts of 60Ni formed?
39.
HP: Accelerated radioactive decay began at the onset of the flood, not only in the fluttering crust but in the pounding and crushing of pillars. As explained on page 340, iron was a common element in pillar tips. During the electrical discharges, bremsstrahlung radiation produced a sea of neutrons throughout the crust. Those neutrons converted some stable iron (54Fe, 56Fe, 57Fe, and 58Fe) into 60Fe which, because of accelerated decay, quickly became 60Ni. Days later, pillar fragments were launched from Earth; some became meteorites.
40.
CE: Iron was produced inside stars. A relatively few stars were so massive that they exploded as supernovas and expelled that iron as a gas into interstellar space. A few ten-millionths of that iron was 60Fe. Before the 60Fe could decay, some must have cooled and merged into dense rocks and crystallized. One of those supernovas had to be “stunningly close” to our solar system for the Sun to capture those rocks so they could later fall to Earth as meteorites.144
[Response: How does gas from a supernova explosion, expanding at almost 20,000 miles per second, quickly merge143 into dense rocks drifting in the vacuum of space? Why did a “stunningly close” supernova not distort, burn, or destroy our solar system? Why can’t we see that nearby supernova’s remnant?]
Deuterium ( 2H). How did deuterium (heavy hydrogen) form, and why is its concentration in comets twice as great as in Earth’s oceans and 20–100 times greater than in interstellar space and the solar system as a whole?
41.
HP: Deuterium formed when the subterranean water absorbed a sea of fast neutrons during the early weeks of the flood. (Powerful bremsstrahlung radiation produces free neutrons, as explained beginning on page 390.) Comets later formed from some of the deuterium-rich water that was launched from Earth by the fountains of the great deep. Traces of that deuterium have been found on the Moon. [See Endnote 76 on page 334.] Most of the deuterium-rich, subterranean water mixed about 50–50 with Earth’s surface waters to give us the high deuterium concentrations we have on Earth today. Meteorites are also rich in deuterium.145
42.
CE: The big bang produced deuterium 3–20 minutes after the universe began, 13.8-billion years ago. During those early minutes, most deuterium was consumed in forming helium. Billions of years later, deuterium that ended up in stars was destroyed. Some deuterium must have escaped that destruction, because comets and Earth have so much deuterium.
Oxygen-18 ( 18O). What is the origin of 18O and why is it concentrated in and around large salt deposits?
43.
HP: Before the flood, the supercritical subterranean water steadily “out-salted” thick layers of water-saturated minerals onto the chamber floor. This included salt crystals (NaCl). [See Endnote 53 on page 141.] The water trapped between those salt crystals absorbed many neutrons during the early weeks of the flood. Later, some of those salt deposits (including their trapped waters) were swept up to the Earth’s surface as thick deposits or rose from the “mother salt layer” as salt domes. Therefore, water in and near thick salt deposits is rich in 18O.
PREDICTION 52: Comets will be found to be rich in 18O.
44.
CE: Presumably, 18O was produced before the Earth evolved. But why 18O is concentrated around large salt deposits is unknown (if the measurements are correct).
Lineaments. How did lineaments form?
45.
HP: Because rocks are weak in tension, fluttering hydroplates sometimes cracked along their convex surfaces when they arched up. This is why lineaments are generally straight cracks, dozens of miles long, parallel to a few directions, found all over the Earth, and show no slippage along the cracks. (Faults show slippage.) Powerful stresses probably converted some long, deep lineaments into faults that produce earthquakes.
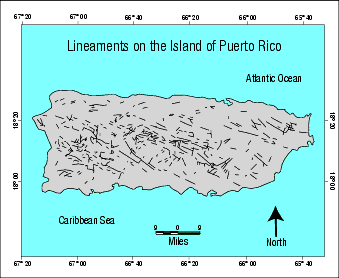
Figure 220: Lineaments. Lineaments are virtually impossible to detect from the ground, because they usually have no vertical or horizontal offsets. On Puerto Rico, the U.S. Geological Survey detected lineament segments (shown as thin black lines) using computer-processed data from side-looking airborne radar, flown 5 miles above the ground. Radar reflections from rock fractures were then digitized and processed by computers that “connected the dots.” The 636 lineaments identified were up to 15 miles in length. The absence of lineaments near coastlines is attributed to thick deposits of recent sediments that scattered the radar signals.146

PREDICTION 53: A positive correlation will be found between lineament concentrations and earthquakes.
46.
CE: While we can’t be sure what produced lineaments, two possibilities have been discussed.
We may speculate about their [lineament] origins. One widely suggested hypothesis is that they reflect continuing flexure of the crust in response to the tidal cycles. ... Another view is that the fractures may stem from subtle back-and-forth tectonic tilting of the crust as it responds to gentle upwarping and downwarping on a regional basis, although the cycles of back-and-forth tilting would necessarily be vastly longer than the twice-daily cycle of the tides.147
[Response: No one has observed rocks breaking because of tides or back-and-forth tilting.]
Cold Mars. The Mars Reconnaissance Orbiter has shown that the Martian polar crust is so rigid that seasonally shifting loads of ice at the poles produce little flexure. This implies that Mars’ interior is extremely cold and has experienced surprisingly little radioactive decay.148 (The evidence explained in "Mountains of Venus" on page 31 shows that the interior of Venus is also cold.)
47.
HP: The inner Earth is hot, because the flood produced large-scale movements, frictional heating, electrical activity, and radioactivity within the Earth. Similar events never happened on Mars or Venus, so the interiors of Mars and Venus should be colder.
48.
CE: The solar system formed from a swirling dust cloud containing heavy radioisotopes billions of years ago. Therefore, with further measurements, Mars’ interior will be shown to be hot, similar to Earth’s.
Distant Chemical Elements. Stars and galaxies 12.9 billion light-years away contain chemical elements heavier than hydrogen, helium, lithium—and nickel. If those elements evolved, it must have happened within 0.8-billion years after the big bang (13.8-billion years ago) in order for their light to reach us. This is extremely fast, based on the steps required for chemical evolution. [See “How Old Do Evolutionists Say the Universe Is?” on page 461.]
49.
HP: Almost all chemical elements were created at the beginning, not just hydrogen, helium, and lithium. [See "Heavy Elements" on page 33.]
50.
CE: If the first stars to evolve were somehow extremely large, they would have exploded as supernovas in only a few tens of millions of years. That debris could then have formed second-generation stars containing these heavier chemical elements—all within 0.8-billion years. This would allow the 12.9-billion years needed for their light to reach us.
The Salt-Oil Connection. Why are massive salt deposits often found near large oil fields? Why is Brazil’s Tupi oil field below 6,600 feet of salt and 4.8 miles below the surface of the Atlantic Ocean? How did the Gulf of Mexico and Mediterranean form?
51.
HP: See "Oil—and Mountains of Salt—All in the Right Places" on page 396, and Figure 217 on page 397.
52.
CE: Many unusual things could happen over billions of years. The massive layers of salt under the floors of the Gulf of Mexico and Mediterranean Sea may indicate that those bodies of water evaporated after they were cut off from the Atlantic Ocean.
[Response: If that happened, it would have to be repeated 8–10 times to produce the thick salt layers that are under those sea floors. Highly unlikely! But even if all that evaporation occurred 8–10 times, why didn’t each refilling dissolve the precipitated salt and wash it into the Atlantic Ocean? See Endnote 31 on page 139.]
Rising Himalayas. How were sediments mixed so uniformly and steadily (over 3,200,000,000 years) in a 1,250-mile-wide band (thousands of feet thick) at the southwestern base of the Himalayas ?
53.
HP: Toward the end of the flood, the compression event pushed up the Himalayas in hours. The overlying flood waters rushed off the rising peaks in all directions, carrying well-mixed, deeply-eroded sediments. In that brief time, the compression event and the resulting electrical activity produced the radioactive decay products that some erroneously believe have always been produced at today’s extremely slow rate.
54.
CE: “Well-mixed sediments were dispersed across at least 2000 km [1,250 miles] of the northern Indian margin. ... The great distances of sediment transport and high degree of mixing of detrital zircon ages are extraordinary, and they may be attributed to a combination of widespread orogenesis associated with the assembly of Gondwana, the equatorial position of continents, potent chemical weathering, and sediment dispersal across a nonvegetated landscape.”149
[Response: This explanation may sound scientific, but is vague and speculative. Furthermore, such “extraordinary” mixing could not have gone on for 3.2-billion years—a vast age based on evolutionary assumptions.]

Forming Heavy Nuclei. How do nuclei merge?
55.
HP: Both shock collapse and the Z-pinch produce extreme compression in plasmas that can overcome the repelling (Coulomb) forces of other nuclei. When two nuclei are close enough, the strong force pulls them together. If the merged nucleus is not at the bottom of the valley of stability, it will decay or fission.
It is a mistake to think that fusion requires high temperatures (>108 K) for long times over large, stellarlike volumes. As the Ukrainian experiments have shown, with small amounts of energy, significant fusion (and fission) can occur in 10-8 second with a self-focused (Z-pinched) electron beam in a high-density plasma.116
56.
CE: Supernovas provide the high temperatures and velocities needed for lighter nuclei to penetrate Coulomb barriers. Those temperatures must be hundreds of times greater than temperatures inside stars, so most chemical elements (those heavier than 60 AMU) cannot form on Earth or inside stable stars.
In 1957, E. Margaret Burbidge, Geoffrey R. Burbidge, William A. Fowler, and Fred Hoyle published a famous paper in which they proposed how supernovas produce all the heavy chemical elements between iron and uranium.151
[Response: See the bolded “Response” on page 400.]
Many supernovas have been seen with powerful telescopes and instruments that can identify the elements and isotopes actually produced. So many elements and isotopes are missing that the supernova explanation must be reexamined.114
6Li, 9Be, 10B, and 11B. Why do we have these light, fragile isotopes on Earth if small impacts will fragment them?
57.
HP: Light, fragile isotopes are too fragile to be created by impacts at the atomic level. Either they were created at the beginning or were produced by extreme compression (shock collapse and the Z-pinch).
Yes, in gases and plasmas, high temperatures produce high particle velocities which might allow nuclei to penetrate the Coulomb barrier. However, if those velocities are slightly larger than necessary, impacted 6Li, 9Be, 10B, and 11B nuclei will fragment. Therefore, high temperatures, instead of fusing those nuclei together, will destroy them.23
58.
CE: Some 6Li, 9Be, 10B, and 11B might be explained by interstellar cosmic rays colliding with carbon, nitrogen, and oxygen, producing 6Li, 9Be, 10B, and 11B fragments.
[Response: Studies of the abundances of these elements and isotopes in stars are inconsistent with this means of producing 6Li, 9Be, 10B, and 11B.152]